Average Atomic Mass Of Nitrogen

Nitrogen is a colourless, odourless unreactive gas that forms most 78% of the earth'due south atmosphere. Liquid nitrogen (made by distilling liquid air) boils at 77.4 kelvins (−195.8°C) and is used as a coolant.
Summary
| Element | Nitrogen |
| Atomic number | 7 |
| Atomic mass [amu] | 14.0067 |
| Atomic mass [pm] | 71 |
| Density at STP [g/cm3] | 0.00125 |
| Number of protons | vii |
| Number of neutrons (typical isotopes) | 14; fifteen |
| Number of electrons | 7 |
| Electron configuration | [He] 2s2 2p3 |
| Oxidation states | +1,2,3,four,5/-i,2,3 |
| Electron analogousness [kJ/mol] | vii |
| Electronegativity [Pauling scale] | 3.04 |
| Start ionization energy [eV] | xiv.5341 |
Diminutive Number – Protons, Electrons and Neutrons in Nitrogen
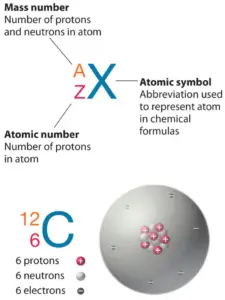 Nitrogen is a chemical chemical element with atomic numbervii which means there are vii protons in its nucleus. Total number of protons in the nucleus is called thediminutive number of the atom and is given thesymbol Z. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals toone,602 x ten-19 coulombs.
Nitrogen is a chemical chemical element with atomic numbervii which means there are vii protons in its nucleus. Total number of protons in the nucleus is called thediminutive number of the atom and is given thesymbol Z. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals toone,602 x ten-19 coulombs.
The full number of neutrons in the nucleus of an atom is chosen theneutron number of the atom and is given thesymbol N. Neutron number plus atomic number equals diminutive mass number:N+Z=A. The departure between the neutron number and the diminutive number is known as theneutron excess: D = N – Z = A – 2Z.
For stable elements, there is usually a variety of stable isotopes.Isotopes are nuclides that have the same atomic number and are therefore the aforementioned element, but differ in the number of neutrons. Mass numbers of typical isotopes ofNitrogen are14; xv.
Atomic Mass of Nitrogen
Atomic mass ofNitrogen is14.0067 u.
The atomic mass is the mass of an cantlet. The diminutive mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The diminutive mass is carried by the diminutive nucleus, which occupies only about 10-12of the total volume of the cantlet or less, merely information technology contains all the positive accuse and at to the lowest degree 99.95% of the total mass of the atom. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance.
Atomic Radius of Nitrogen
The atomic radius ofNitrogenatom is 71pm (covalent radius).
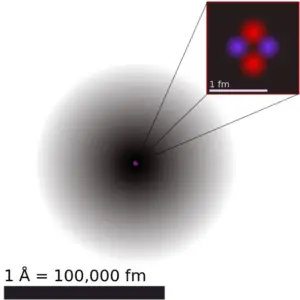
It must be noted, atoms lack a well-defined outer boundary. The diminutive radius of a chemical element is a measure of the distance out to which the electron deject extends from the nucleus. Notwithstanding, this assumes the atom to exhibit a spherical shape, which is just obeyed for atoms in vacuum or free space. Therefore, there are various not-equivalent definitions of atomic radius.
Electrons and Electron Configuration
The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. Therefore, the number of electrons in neutral cantlet ofNitrogenis7. Each electron is influenced by the electric fields produced by the positive nuclear charge and the other (Z – 1) negative electrons in the atom.
Since the number of electrons and their arrangement are responsible for the chemic behavior of atoms, thediminutive number identifies the various chemical elements. The configuration of these electrons follows from the principles of quantum mechanics. The number of electrons in each element'south electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. In the periodic tabular array, the elements are listed in social club of increasing diminutive number Z.
Electron configuration ofNitrogenis[He] 2s2 2p3.
Possible oxidation states are+1,two,3,4,v/-1,two,3.
Density of Nitrogen
Density ofNitrogenis0.00125g/cm3 .
Typical densities of diverse substances are at atmospheric pressure.
Density is defined as themass per unit volume. It is anintensive property, which is mathematically defined as mass divided by volume:
ρ = grand/V
Atomic Masses of Elements
Diminutive Radii of Elements
Densities of Elements


Electron Affinity – Nitrogen
Electron affinity ofNitrogen is7 kJ/mol.
In chemistry and diminutive physics, theelectron affinity of an atom or molecule is defined every bit:
the change in energy (in kJ/mole) of a neutral cantlet or molecule (in the gaseous stage) when an electron is added to the atom to grade a negative ion.
X + e– → 10– + energy Affinity = – ∆H
In other words, it tin can be expressed as the neutral atom'southwardlikelihood of gaining an electron. Note that, ionization energies measure the tendency of a neutral atom to resist the loss of electrons. Electron affinities are more hard to measure than ionization energies.
Electronegativity of Nitrogen
Electronegativity ofNitrogen isthree.04.
Electronegativity, symbol χ, is a chemical holding that describes the trend of an cantlet to concenter electrons towards this atom. For this purposes, adimensionless quantity thePauling scale, symbol χ, is the most normally used.
The electronegativity of Nitrogen is: χ = 3.04
Kickoff Ionization Energy of Nitrogen
Get-go Ionization Free energy of Nitrogen is 14.5341 eV.
Ionization free energy, also chosen ionization potential, is the energy necessary to remove an electron from the neutral cantlet.
X + energy → Ten+ + east−
where Ten is any cantlet or molecule capable of existence ionized, X+ is that atom or molecule with an electron removed (positive ion), and e− is the removed electron.
A Nitrogen atom, for example, requires the following ionization energy to remove the outermost electron.
Due north + IE → Due north+ + e− IE = 14.5341 eV
Electronegativity of Elements

Ionization Free energy of Elements

Source: www.luciteria.com
Other properties of Nitrogen
Average Atomic Mass Of Nitrogen,
Source: https://material-properties.org/Nitrogen-periodic-table-atomic-number-mass-radius-density/
Posted by: alcazarsarlizies.blogspot.com


0 Response to "Average Atomic Mass Of Nitrogen"
Post a Comment